Tata Elxsi at MedTech Summit Berlin 2026
JW Marriott Hotel, Berlin, Germany | 15 - 18 June 2026
Book a Meeting

Tata Elxsi introduces ViTel™, a Material Intelligence Solution, Powered by Viridium AI
For smarter material insights, compliance, & faster decision-making
Read Press Release

Tata Elxsi and OpenAna Launch AnaTel™, an AI-Native Software Development Platform
AnaTel™ helps medical device and SaMD companies accelerate software development, generate regulatory-grade documentation, and reduce development cycle times
Read Press Release

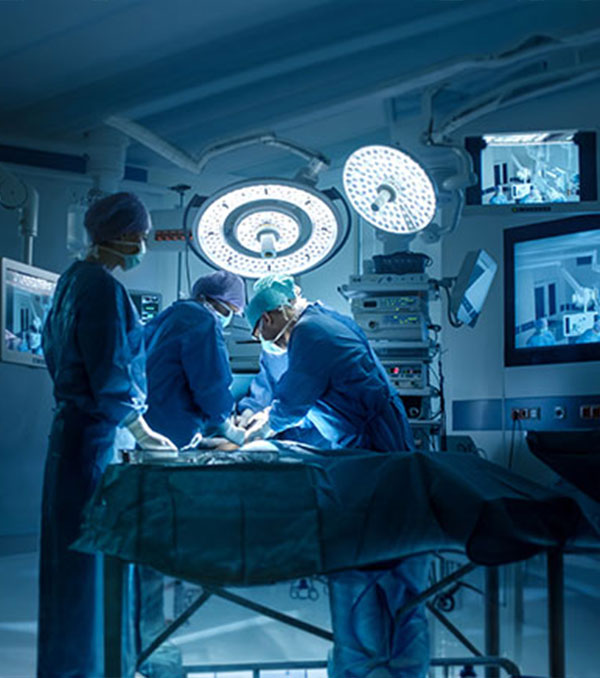
Surgical robots are Not New. Intelligent Ones are
For more than a decade, robotic systems have been a fixture in advanced operating rooms, enabling surgeons to perform minimally invasive procedures with greater precision and control.
Read Article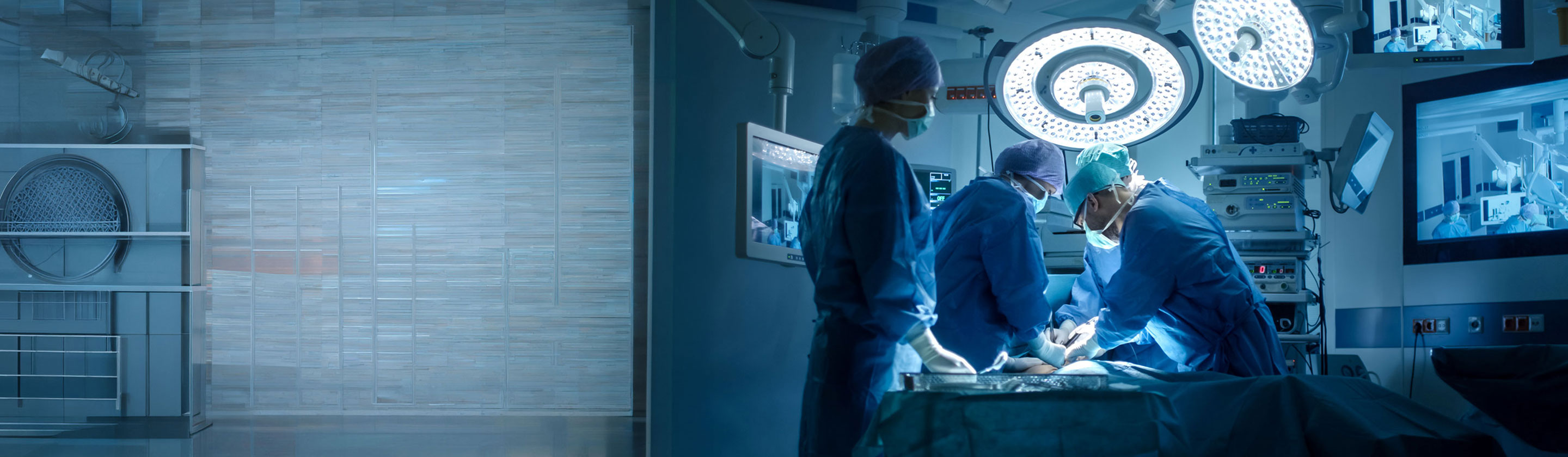
Healthcare & Wellness Trends Outlook 2026
What’s next in health, innovation, and wellbeing? Expert Analysis by Anand Jayaraman Payer & Provider Segment Head – Healthcare and Life Sciences, Tata Elxsi
Watch Now
Charting the Future of Radiology
Distinguish genuine innovation from hype, operationalize compliance for SaMD and AI/ML-enabled devices to continuously improve safety, performance, and trust.
Watch NowTransforming the Healthcare and Life Sciences Industry
The healthcare and life sciences industry is undergoing a profound evolution, driven by a focus on affordability, accessibility, and quality care. Organizations are leveraging cutting-edge technologies like Gen AI, robotics, miniaturization, and connected care ecosystems to optimize operations and enhance patient outcomes.
At Tata Elxsi, we're at the forefront of this transformation, collaborating with healthcare leaders to unlock new opportunities and drive meaningful impact. Together, we are shaping a future where innovation and technology empower healthier lives for all while ensuring compliance with stringent regulatory standards.
A Portable Diagnostic Platform Offering Affordable and Accessible Care
Business Challenge
Organizations in healthcare and life sciences face mounting pressures to innovate quickly while meeting stringent regulatory requirements and addressing rising costs. Rapid technological advancements demand agile adoption of digital healthcare solutions and seamless integration of legacy systems. Achieving patient-centric outcomes amid data security concerns and interoperability challenges forms the crux of modern business imperatives in this complex, evolving landscape.
Healthcare and Life Sciences Products & Services
Tata Elxsi offers end-to-end healthcare solutions, from R&D and product engineering to digital strategy, ensuring seamless patient experiences, regulatory compliance, and operational efficiency across the entire healthcare value chain.
Medical Device Design and Engineering Services | Digital Health Engineering | Medical Device Regulatory Compliance | Payers | Providers | ISVs
What's New
Information Hub
-
What are the latest trends shaping the healthcare and life sciences industry?
Emerging digital health solutions, personalized medicine, and patient-centric care models are driving a shift toward value-based outcomes. Artificial intelligence (AI) and machine learning are transforming diagnostic accuracy, while advanced biosensors enable continuous patient monitoring. Blockchain supports secure data sharing and improved interoperability, ensuring transparency in clinical trials. Telemedicine and remote care are expanding access to healthcare services globally, driven by consumer demand and regulatory changes. In parallel, sustainability and green healthcare initiatives are becoming critical, reflecting broader societal priorities. Collectively, these trends redefine the scope of healthcare innovation and the pace of its delivery.
-
What are key considerations in medical device innovation today?
Successful medical device innovation demands a convergence of human-centered design, regulatory foresight, and technology scalability. Companies must factor in real-world usability, clinical validation, cybersecurity, and compliance with evolving standards such as FDA, EU MDR, and ISO 13485. Partnering with the right medical device engineering and design teams ensures faster, safer, and more sustainable product launches.
-
What role does digital transformation play in improving patient outcomes?
Digital transformation is fundamental to modern healthcare as it integrates AI, analytics, and cloud-based platforms for real-time insights and informed decision-making. By connecting healthcare providers, patients, and payers through telemedicine, remote monitoring, and data-driven diagnostics, it reduces treatment delays and human errors. This interconnected ecosystem enhances patient engagement, personalizes treatment plans, and promotes preventive care, ultimately leading to better health outcomes. Additionally, digital solutions streamline administrative workflows and improve resource allocation, ensuring healthcare professionals can focus on delivering high-quality, patient-centric services. The result is an agile, scalable infrastructure designed to evolve with emerging needs.
-
How can healthcare organizations leverage data analytics for strategic decision-making?
By harnessing big data from electronic health records, clinical trials, and connected devices, healthcare organizations can uncover insights that inform policy, optimize resource allocation, and improve patient outcomes. Predictive analytics and machine learning models help identify disease patterns, anticipate patient needs, and personalize treatments. This evidence-based approach reduces redundancies, highlights cost-saving opportunities, and fosters innovation in product development and service delivery. Additionally, advanced analytics enable continuous monitoring of key performance indicators, ensuring that strategic decisions align with regulatory standards and quality of care benchmarks, ultimately driving sustainable growth and enhanced patient satisfaction.
-
What does the future of healthcare look like with emerging technologies?
Future healthcare systems will be increasingly patient-centric, fueled by advancements in genomics, AI-driven diagnostics, and extended reality tools for immersive training. Wearable devices and smart implants will capture real-time data, enabling personalized treatment plans and continuous patient monitoring. 5G connectivity will streamline telemedicine services, facilitating remote surgeries and collaborative care across geographies. Simultaneously, data privacy regulations will evolve to safeguard sensitive health information. Overall, these emerging technologies promise to reduce costs, minimize errors, and expand access to quality healthcare, ensuring a collaborative and holistic approach to global well-being.
-
How does Tata Elxsi ensure regulatory compliance in product development?
Tata Elxsi follows established global standards such as ISO 13485, FDA guidelines, and EU MDR requirements to design and develop medical devices and digital health platforms. Our processes incorporate robust risk management strategies, from requirements gathering to verification and validation. We maintain comprehensive documentation to meet global regulatory demands, ensuring traceability and transparency at every stage. By integrating compliance from the earliest design phases and leveraging cross-functional teams with deep domain knowledge, Tata Elxsi helps clients navigate complex requirements. This systematic approach accelerates market entry while minimizing costly non-compliance risks, guaranteeing safe and effective solutions.
-
Why is sustenance engineering critical for medical devices?
Sustenance engineering ensures that legacy and in-market medical devices remain compliant, secure, and supportable. It enables effective obsolescence management, value engineering, and faster adaptation to regulatory changes, extending product life and reducing total cost of ownership. As post-market requirements grow, a strong sustenance strategy is essential.