Attention
This website is best viewed in portrait mode.
ROHS2 compliance for drug delivery device
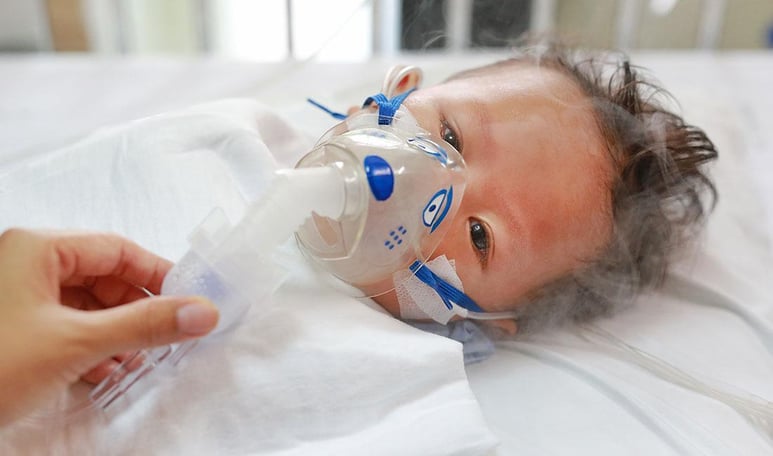
The Customer:
World leader in Nitric Oxide drug delivery system for critical neonatal care
Problem Statement:
Client intended
- Provable reduction of restricted substance to compliant levels (RoHS2 including REACH) in its Nitric Oxide drug delivery device, its accessories and packaging
- Re-engineering of the existing drug delivery system to ensure RoHS2 compliance
- Streamlining the documentation procedure to ensure fast and efficient compilation of data for components and materials used in the device, the accessories, disposables & packaging
- Ensure continued ability to place the “CE” mark through compliance to the MDD in Europe
- Ensure better product reliability through active control of RoHS electronic component lead plating and soldering process
Project Description & Scope:
- Technical assessment of Nitric Oxide drug delivery device
- Detailed study of product in totality usings Bill of Material (BOM), schematics, drawings etc.
- Re-engineering of hardware and mechanical components of Nitric Oxide drug delivery device
- Selection of functionally equivalent RoHS components
- Verification of RoHS compliance certification/material declaration and certificate of each component
- Fabrication of RoHS compliant hardware PCBs
- Functional and integration testing
- EMI/EMC pre-compliance testing
- RoHS documentation required for CE marking
- Drawing the missing components (Electronic & Mechanical) & updating the BOM
Business Benefit:
- Time consuming activity of collecting Certificate of Compliance done by Tata Elxsi’s team
- Redesigning PCB without much involvement of the core design team
- Ensuring order fulfillment and business continuity in EU and expansion into Japan and BRIC markets
Tools Used:
PCB Design
- PADS version 9.5
- Altium version 10
Mechanical Drawings
- SolidWorks x64 Edition professional 2012